Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 3.05⋅107 | ± 2.70⋅106 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -42.72 | ± 0.22 | -10.21 | ± 0.05 |
| ΔH | = | -36.7 | ± 1.9 | -8.77 | ± 0.45 |
| -TΔS | = | -6.0 | ± 1.7 | -1.43 | ± 0.41 |
| J mol-1 K-1 | cal mol-1 K-1 | ||||
| ΔS | = | 20.1 | ± 5.7 | 4.8 | ± 1.4 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Isothermal Titration Calorimetry | ||
| Instrument: | MicroCal VP-ITC | ||
| VCell | = | 1400.0 𝜇L | |
| VSyringe | = | 350.0 𝜇L | |
| Molecule: | syringe | ||
| Partner: | cell | ||
Detailed information about the solvation.
| Solvent System | Complex Mixture |
| Solvents | water |
| Additives | hydrochloric acid |
| pH | 6.0 |
Please find here information about the dataset this interaction is part of.
| Citation: |
D. Guo, Y. Liu, W. M. Nau, V. D. Uzunova, K. I. Assaf, A. I. Lazar, SupraBank 2026, Inclusion of neutral guests by water-soluble macrocyclic hosts – a comparative thermodynamic investigation with cyclodextrins, calixarenes and cucurbiturils (dataset). https://doi.org/10.34804/supra.2021092882 |
| Link: | https://doi.org/10.34804/supra.2021092882 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
D.-S. Guo, V. D. Uzunova, K. I. Assaf, A. I. Lazar, Y. Liu, W. M. Nau, Supramolecular Chemistry 2015, 28, 384–395. |
| Link: | https://doi.org/10.1080/10610278.2015.1105374 |
| Export: | BibTex | RIS | EndNote |
Binding Isotherm Simulations
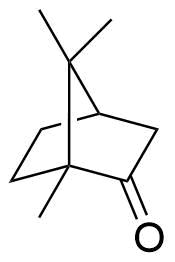
The plot depicts the binding isotherm simulation of a 1:1 interaction of Camphor (6.557377049180327e-07 M) and CB8 (0 — 1.3114754098360655e-06 M).
Please sign in: customize the simulation by signing in to the SupraBank.