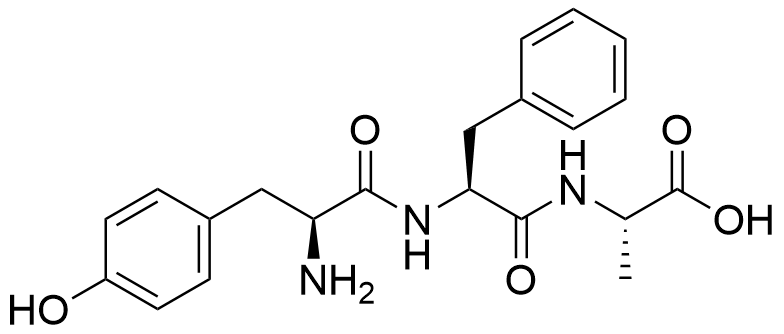
Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 3.45⋅106 | ± 4.80⋅105 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 27.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -37.57 | ± 0.35 | -8.98 | ± 0.08 |
| ΔH | = | -67.36 | ± 0.84 | -16.1 | ± 0.2 |
| -TΔS | = | 30.12 | ± 0.42 | 7.2 | ± 0.1 |
| J mol-1 K-1 | cal mol-1 K-1 | ||||
| ΔS | = | -100.3 | ± 1.4 | -24.0 | ± 0.3 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Isothermal Titration Calorimetry | ||
| Molecule: | syringe | ||
| Partner: | cell | ||
Detailed information about the solvation.
| Solvent System | Buffer System | 10 mM phosphate pH-7.0 |
| Solvents | water | |
| Source of Concentration | ||
| Total concentration | 10.0 mM | |
| pH | 7.0 |
Please find here information about the dataset this interaction is part of.
| Citation: |
A. R. Urbach, L. C. Smith, D. G. Leach, B. E. Blaylock, O. A. Ali, SupraBank 2026, Sequence-Specific, Nanomolar Peptide Binding via Cucurbit[8]uril-Induced Folding and Inclusion of Neighboring Side Chains (dataset). https://doi.org/10.34804/supra.20210928193 |
| Link: | https://doi.org/10.34804/supra.20210928193 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
L. C. Smith, D. G. Leach, B. E. Blaylock, O. A. Ali, A. R. Urbach, J. Am. Chem. Soc. 2015, 137, 3663–3669. |
| Link: | https://doi.org/10.1021/jacs.5b00718 |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
The plot depicts the binding isotherm simulation of a 1:1 interaction of Tyr-Phe-Ala (5.80046403712297e-06 M) and CB8 (0 — 1.160092807424594e-05 M).
Please sign in: customize the simulation by signing in to the SupraBank.