Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 11.0 | ± 2.0 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -5.94 | ± 0.46 | -1.42 | ± 0.11 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Nuclear Magnetic Resonance | ||
| Nucleus | 1H | ||
Detailed information about the solvation.
| Solvent System | Complex Mixture | |
| Solvents | Deuterium Oxide | |
| Additives | Sodium sulfate | 200.0 mM |
| Total concentration | 200.0 mM |
Please find here information about the dataset this interaction is part of.
| Citation: |
K. Bartik, Y. Ho Ko, K. Kim, M. E. Haouaj, M. Luhmer, SupraBank 2026, NMR Investigation of the complexation of neutral guests by cucurbituril (dataset). https://doi.org/10.34804/supra.20210928350 |
| Link: | https://doi.org/10.34804/supra.20210928350 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
M. E. Haouaj, Y. Ho Ko, M. Luhmer, K. Kim, K. Bartik, J. Chem. Soc., Perkin Trans. 2 2001, 2104–2107. |
| Link: | https://doi.org/10.1039/b105535f |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
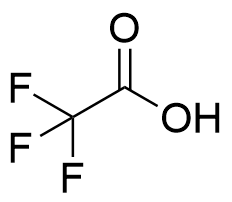
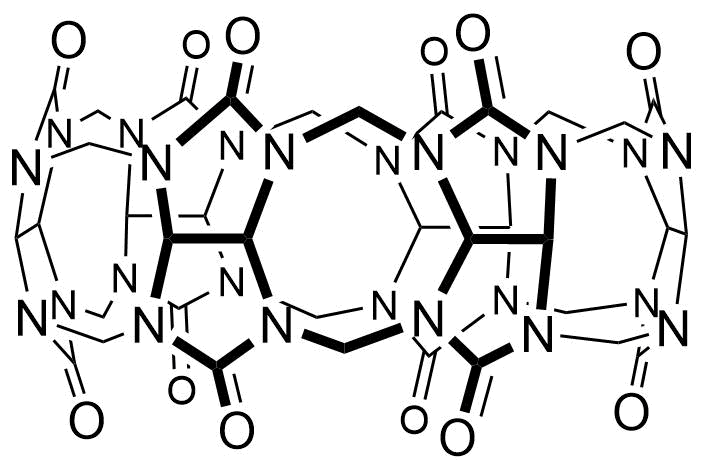
The plot depicts the binding isotherm simulation of a 1:1 interaction of Trifluoroacetic acid (1.8181818181818181 M) and CB6 (0 — 3.6363636363636362 M).
Please sign in: customize the simulation by signing in to the SupraBank.