Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 6.10⋅107 | ± 4.00⋅106 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| kin= | 90000000.0 | ± 9000000.0 | M-1s-1 |
| kout= | 1.47540983606557 | ± 0.2 | s-1 |
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -44.44 | ± 0.16 | -10.62 | ± 0.04 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | |||
| Assay Type: | Direct Binding Assay | |||
| Technique: | Fluorescence | |||
| 𝛌ex | = | 384.0 nm | ||
| 𝛌em | = | 440.0 nm | ||
Detailed information about the solvation.
| Solvent System | Single Solvent |
| Solvent | water |
Please find here information about the dataset this interaction is part of.
| Citation: |
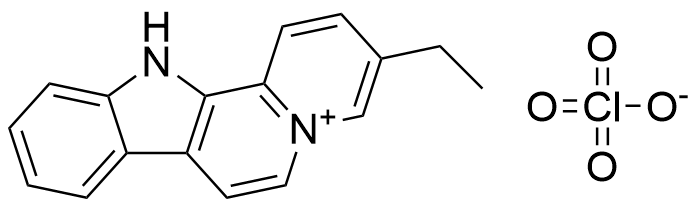
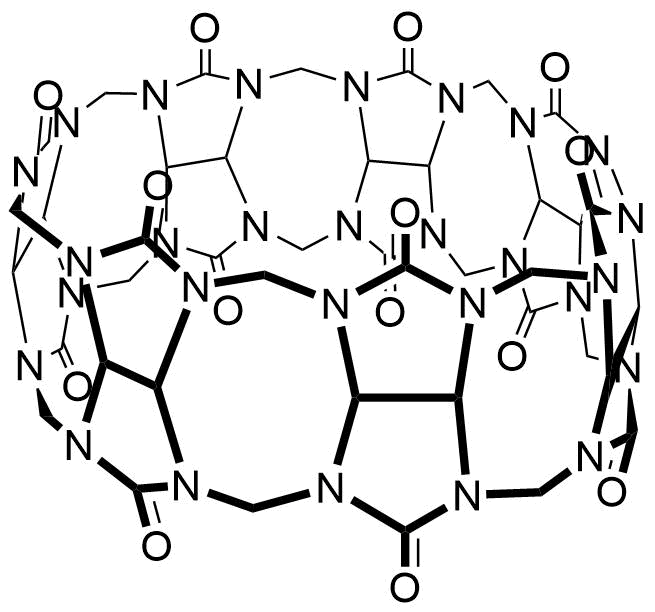
Z. Miskolczy, L. Biczok, I. Jablonkai, SupraBank 2026, Kinetics of the reversible inclusion of flavopereirine in cucurbit[7]uril (dataset). https://doi.org/10.34804/supra.20210928116 |
| Link: | https://doi.org/10.34804/supra.20210928116 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
Z. Miskolczy, L. Biczók, I. Jablonkai, Phys. Chem. Chem. Phys. 2017, 19, 766–773. |
| Link: | https://doi.org/10.1039/C6CP07553C |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
The plot depicts the binding isotherm simulation of a 1:1 interaction of Flavopereirine perchlorate (3.2786885245901637e-07 M) and CB7 (0 — 6.557377049180327e-07 M).
Please sign in: customize the simulation by signing in to the SupraBank.