Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 1880.0 | ± 330.0 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -18.69 | ± 0.44 | -4.47 | ± 0.11 |
| ΔH | = | -17.95 | ± 0.83 | -4.29 | ± 0.2 |
| -TΔS | = | -0.71 | ± 1.3 | -0.17 | ± 0.31 |
| J mol-1 K-1 | cal mol-1 K-1 | ||||
| ΔS | = | 2.4 | ± 4.4 | 0.6 | ± 1.0 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Isothermal Titration Calorimetry | ||
| Molecule: | syringe | ||
| Partner: | cell | ||
Detailed information about the solvation.
| Solvent System | Buffer System | 50 mM phosphate buffer with NaCl & urea, pH 8.6 |
| Solvents | water | |
| Additives | urea | 8000.0 mM |
| sodium chloride | 100.0 mM | |
| phosphate | 50.0 mM | |
| Source of Concentration | real | |
| Total concentration | 8150.0 mM | |
| pH | 8.6 |
Please find here information about the dataset this interaction is part of.
| Citation: |
A. E. Kaifer, L. A. Godínez, L. Schwartz, C. M. Criss, SupraBank 2026, Thermodynamic Studies on the Cyclodextrin Complexation of Aromatic and Aliphatic Guests in Water and Water−Urea Mixtures. Experimental Evidence for the Interaction of Urea with Arene Surfaces (dataset). https://doi.org/10.34804/supra.2021092880 |
| Link: | https://doi.org/10.34804/supra.2021092880 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
L. A. Godínez, L. Schwartz, C. M. Criss, A. E. Kaifer, J. Phys. Chem. B 1997, 101, 3376–3380. |
| Link: | https://doi.org/10.1021/jp970359i |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
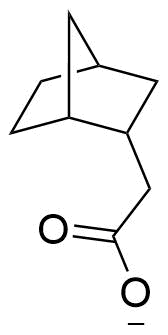
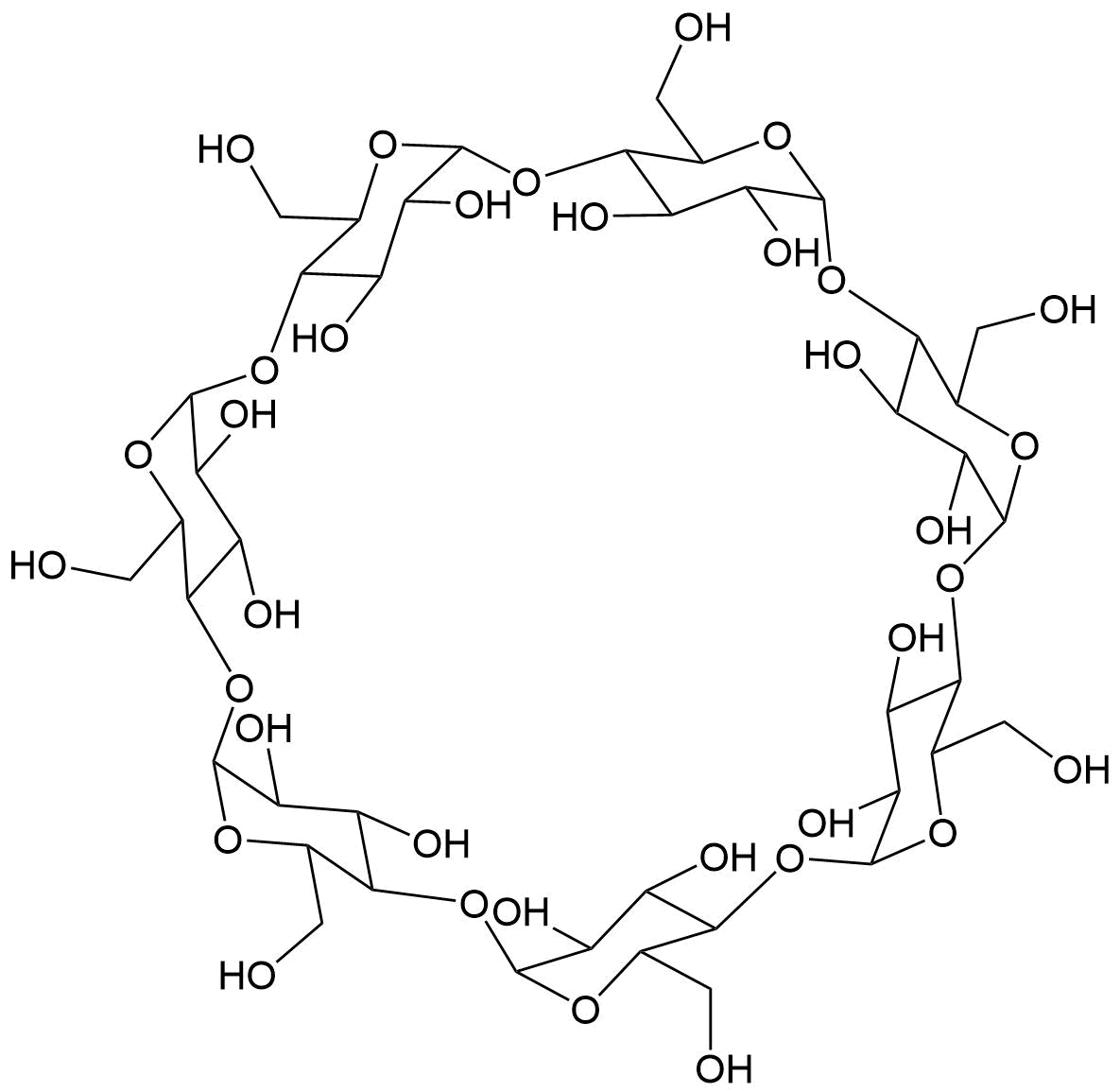
The plot depicts the binding isotherm simulation of a 1:1 interaction of bicyclo[2.2.1]heptan-2-yl)acetate (0.010638297872340425 M) and β-CD (0 — 0.02127659574468085 M).
Please sign in: customize the simulation by signing in to the SupraBank.