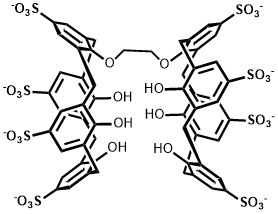
Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 4.27⋅105 | ± 1000.0 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -32.14 | ± 0.01 | -7.68 | ± 0.0 |
| ΔH | = | -39.47 | ± 0.02 | -9.43 | ± 0.0 |
| -TΔS | = | 7.36 | ± 0.01 | 1.76 | ± 0.0 |
| J mol-1 K-1 | cal mol-1 K-1 | ||||
| ΔS | = | -24.7 | ± 0.0 | -5.9 | ± 0.0 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Isothermal Titration Calorimetry | ||
| Molecule: | syringe | ||
| Partner: | syringe | ||
Detailed information about the solvation.
| Solvent System | Buffer System | phosphate pH-7.2 |
| Solvents | water | |
| Source of Concentration | ||
| pH | 7.2 |
Please find here information about the dataset this interaction is part of.
| Citation: |
D. Guo, H. Qian, Y. Liu, H. Zhang, S. Chen, SupraBank 2026, Electrochemical stimulus-responsive supramolecular polymer based on sulfonatocalixarene and viologen dimers (dataset). https://doi.org/10.34804/supra.20210928282 |
| Link: | https://doi.org/10.34804/supra.20210928282 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
D.-S. Guo, S. Chen, H. Qian, H.-Q. Zhang, Y. Liu, Chem. Commun. 2010, 46, 2620. |
| Link: | https://doi.org/10.1039/b925157j |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
The plot depicts the binding isotherm simulation of a 1:1 interaction of bis-MV4+ (4.68384074941452e-05 M) and bis-sCx4 (0 — 9.36768149882904e-05 M).
Please sign in: customize the simulation by signing in to the SupraBank.