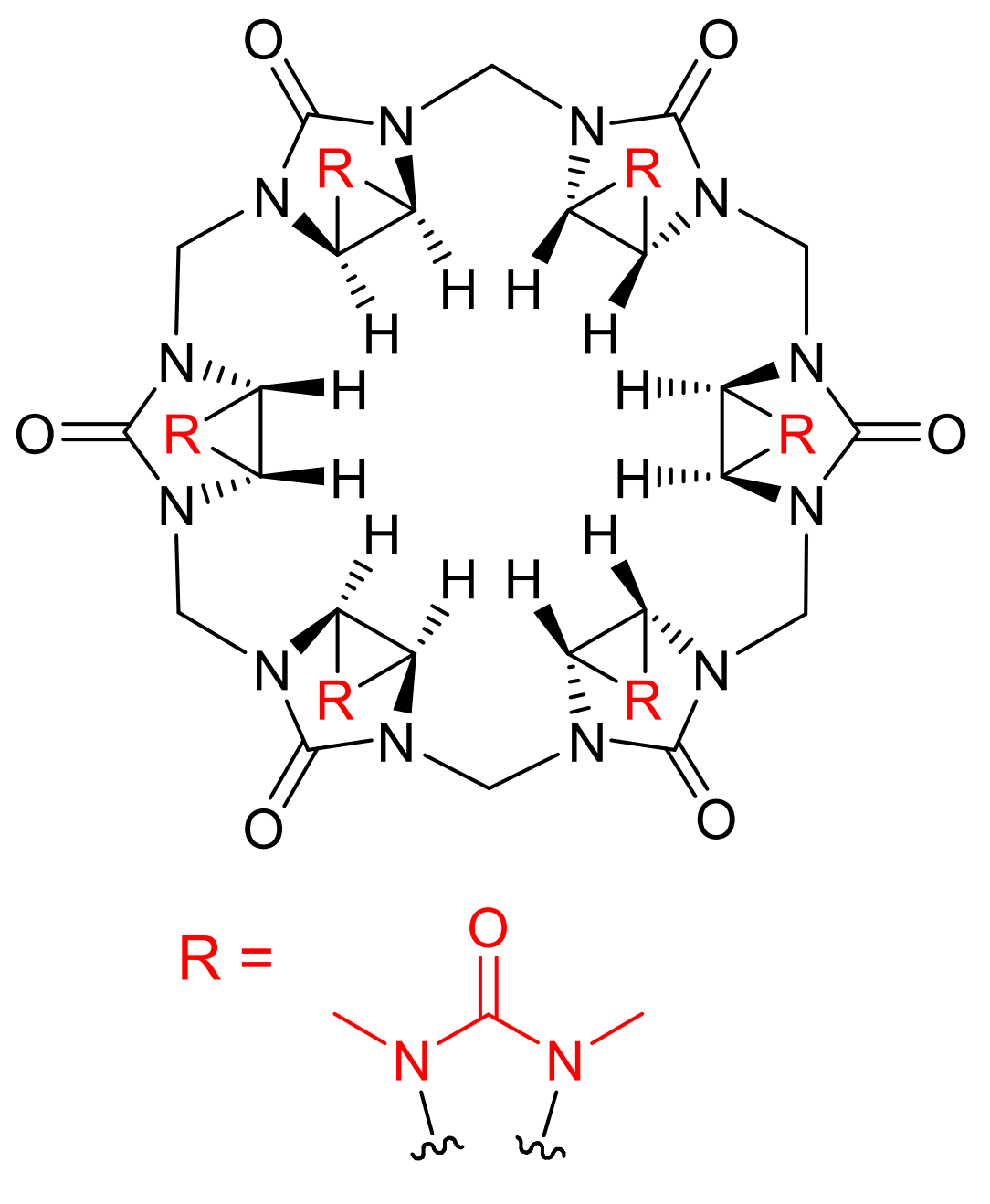
Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 2.09⋅104 | M-1 | |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -24.66 | -5.89 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Nuclear Magnetic Resonance | ||
Detailed information about the solvation.
| Solvent System | Complex Mixture | |
| Solvents | Acetonitrile-d3 | 50.0 % |
| Deuterium Oxide | 50.0 % | |
Please find here information about the dataset this interaction is part of.
| Citation: |
M. Singh, E. Solel, E. Keinan, O. Reany, SupraBank 2026, Dual-Functional Semithiobambusurils (dataset). https://doi.org/10.34804/supra.20210928136 |
| Link: | https://doi.org/10.34804/supra.20210928136 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
M. Singh, E. Solel, E. Keinan, O. Reany, Chem. Eur. J. 2014, 21, 536–540. |
| Link: | https://doi.org/10.1002/chem.201404210 |
| Export: | BibTex | RIS | EndNote |
Binding Isotherm Simulations
The plot depicts the binding isotherm simulation of a 1:1 interaction of Tetrafluoroborate (0.0009573958831977022 M) and Me12BU (0 — 0.0019147917663954045 M).
Please sign in: customize the simulation by signing in to the SupraBank.