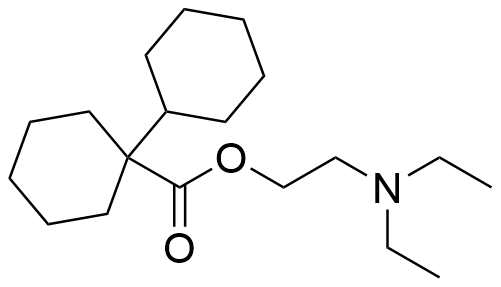
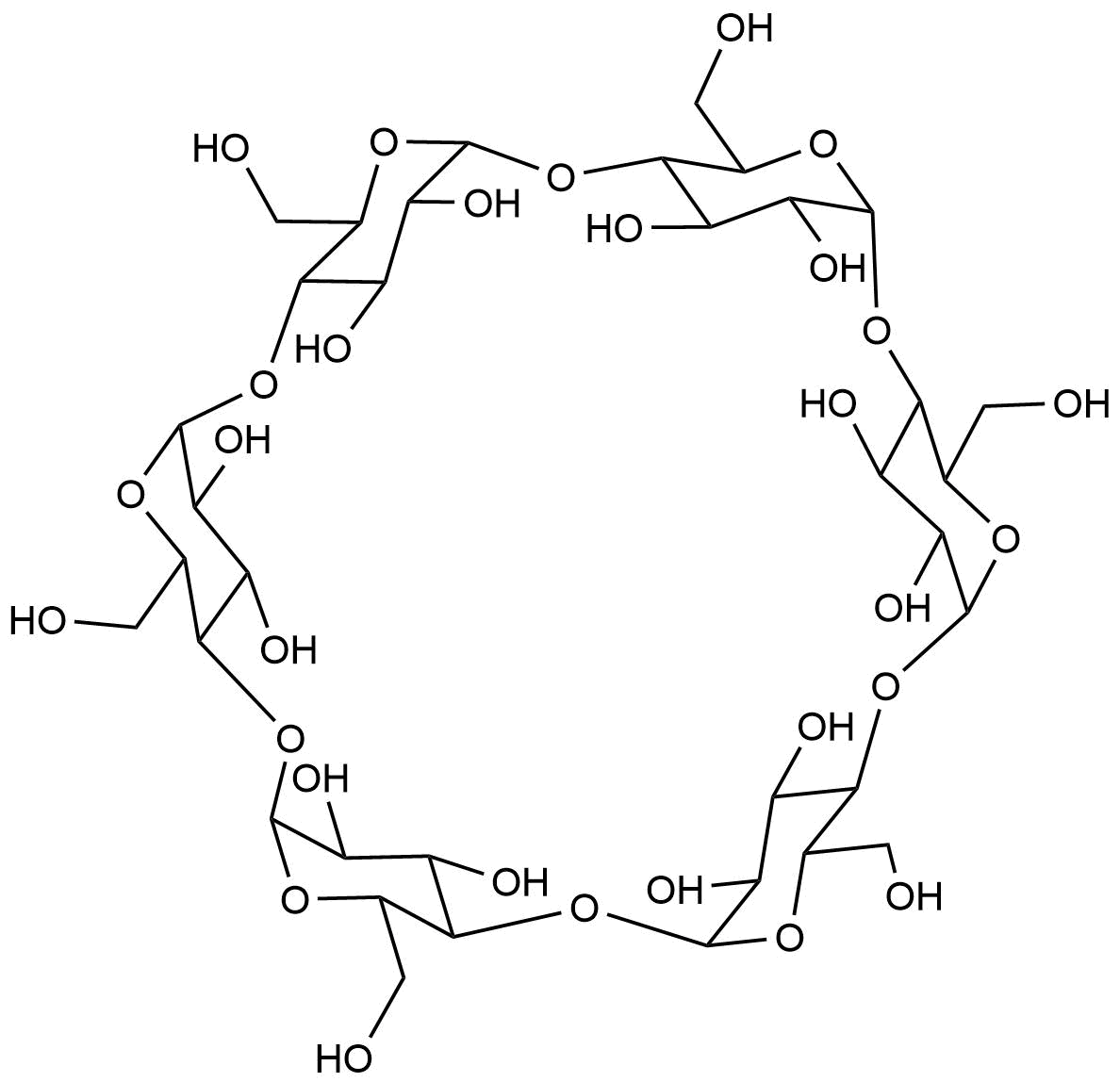
Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 280.0 | ± 30.0 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -13.97 | ± 0.27 | -3.34 | ± 0.06 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Potentiometry | ||
Detailed information about the solvation.
| Solvent System | Complex Mixture | |
| Solvents | water | |
| water | ||
| Additives | sodium chloride | 10.0 mM |
Please find here information about the dataset this interaction is part of.
| Citation: |
N. Takisawa, D. G. Hall, E. Wyn-Jones, P. Brown, SupraBank 2026, The construction and characteristics of drug-selective electrodes. Applications for the determination of complexation constants of inclusion complexes with α- and β-cyclodextrins including a kinetic study (dataset). https://doi.org/10.34804/supra.20210928323 |
| Link: | https://doi.org/10.34804/supra.20210928323 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
N. Takisawa, D. G. Hall, E. Wyn-Jones, P. Brown, J. Chem. Soc., Faraday Trans. 1 1988, 84, 3059. |
| Link: | https://doi.org/10.1039/f19888403059 |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
The plot depicts the binding isotherm simulation of a 1:1 interaction of dicyclomine (0.07142857142857142 M) and α-CD (0 — 0.14285714285714285 M).
Please sign in: customize the simulation by signing in to the SupraBank.