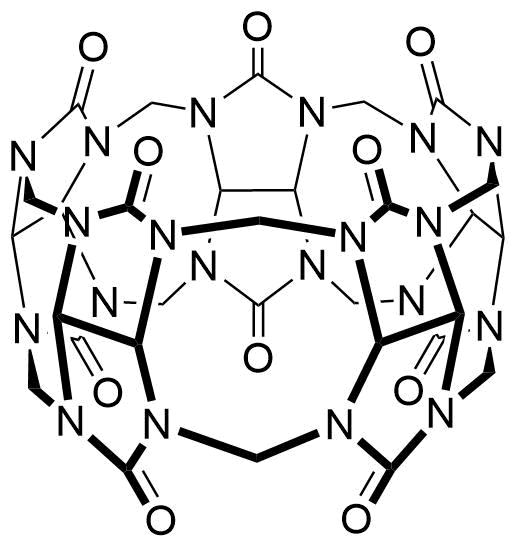
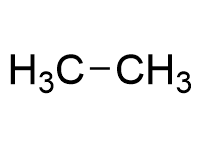Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 87.0 | ± 1.0 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -11.07 | ± 0.03 | -2.65 | ± 0.01 |
- Comment
- single point measurements under consideration of gas solubities in water; no titrations
These are the specifications of the determination of the experimental results.
| Detection Method: | Competitive | ||
| Assay Type: | Competitive Binding Assay | ||
| Technique: | Nuclear Magnetic Resonance | ||
| Nucleus | H-1 | ||
Detailed information about the solvation.
| Solvent System | Single Solvent |
| Solvent | Deuterium Oxide |
| pH | 7.0 |
Please find here information about the dataset this interaction is part of.
| Citation: |
W. M. Nau, F. Biedermann, A. De Simone, S. He, N. Vankova, L. Zhechkov, T. Heine, R. E. Hoffman, T. T. Duignan, SupraBank 2026, Cavitation energies can outperform dispersion interactions (dataset). https://doi.org/10.34804/supra.20220701455 |
| Link: | https://doi.org/10.34804/supra.20220701455 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
S. He, F. Biedermann, N. Vankova, L. Zhechkov, T. Heine, R. E. Hoffman, A. De Simone, T. T. Duignan, W. M. Nau, Nature Chem 2018, 10, 1252–1257. |
| Link: | https://doi.org/10.1038/s41557-018-0146-0 |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
The plot depicts the binding isotherm simulation of a 1:1 interaction of He (0.22988505747126436 M) and CB5 (0 — 0.45977011494252873 M).
Please sign in: customize the simulation by signing in to the SupraBank.