Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 3.60⋅109 | ± 5.00⋅108 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -54.55 | ± 0.35 | -13.04 | ± 0.08 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Competitive | ||
| Assay Type: | Competitive Binding Assay | ||
| Technique: | Absorbance | ||
Detailed information about the solvation.
| Solvent System | Buffer System | 20 mM phosphate pH-7.4 |
| Solvents | water | |
| Additives | Sodium dihydrog... | 20.0 mM |
| Source of Concentration | ||
| Total concentration | 20.0 mM | |
| pH | 7.4 |
Please find here information about the dataset this interaction is part of.
| Citation: |
L. Isaacs, P. Y. Zavalij, D. Ma, SupraBank 2025, Acyclic Cucurbit[n]uril Congeners Are High Affinity Hosts (dataset). https://doi.org/10.34804/supra.20240305511 |
| Link: | https://doi.org/10.34804/supra.20240305511 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
D. Ma, P. Y. Zavalij, L. Isaacs, J. Org. Chem. 2010, 75, 4786–4795. |
| Link: | https://doi.org/10.1021/jo100760g |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
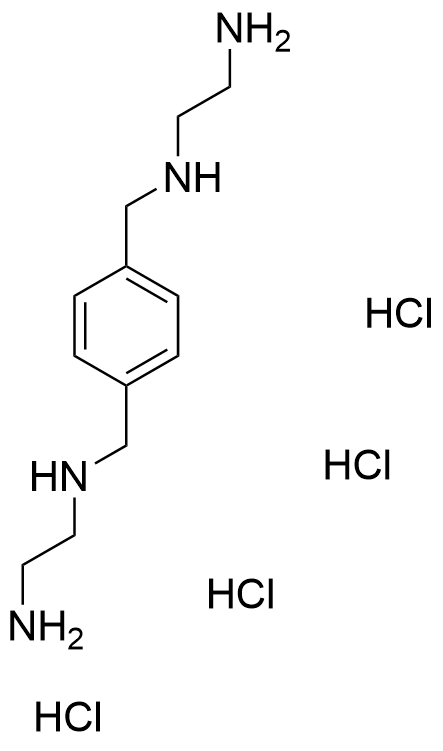
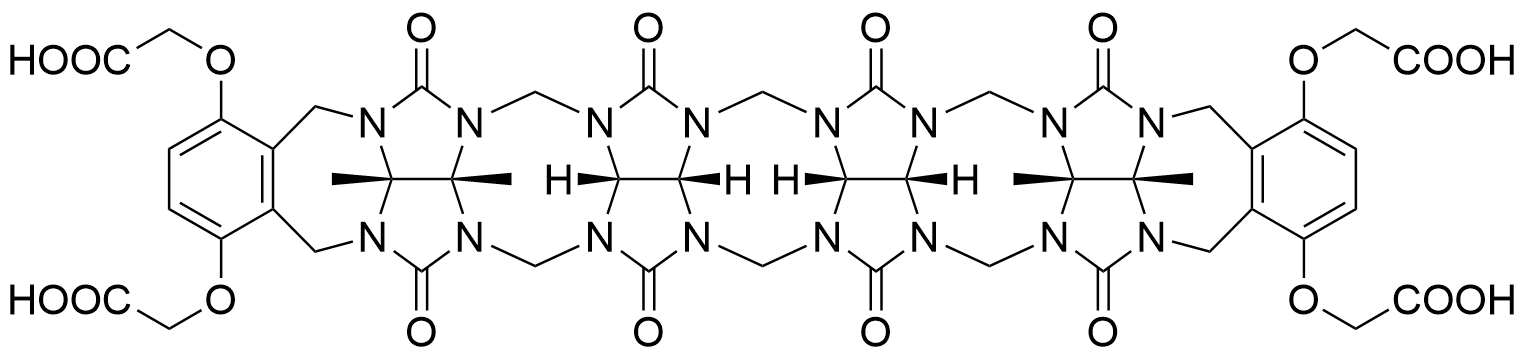
The plot depicts the binding isotherm simulation of a 1:1 interaction of N1,N1'-(1,4-phenylenebis(methylene))bis(ethane-1,2-diamine) tetrahydrochloride (5.555555555555556e-09 M) and CB5a (0 — 1.1111111111111112e-08 M).
Please sign in: customize the simulation by signing in to the SupraBank.