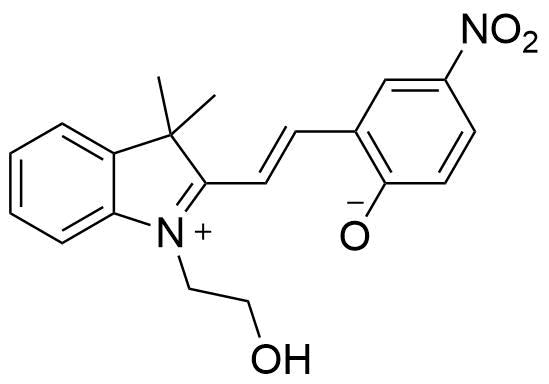
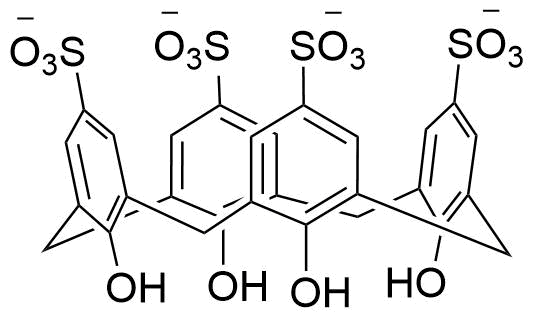
Binding Properties
| 𝜈 | Molecule 1 : 1 Host | ||
| Ka = | 3500.0 | ± 400.0 | M-1 |
| Kd = | |||
| logKa = | |||
| T | 25.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -20.23 | ± 0.28 | -4.84 | ± 0.07 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | |||
| Assay Type: | Direct Binding Assay | |||
| Technique: | Fluorescence | |||
| 𝛌ex | = | 502.0 nm | ||
| 𝛌em | = | 632.0 nm | ||
Detailed information about the solvation.
| Solvent System | Complex Mixture |
| Solvents | water |
| Additives | Potassium hydro... |
| pH | 7.8 |
Please find here information about the dataset this interaction is part of.
| Citation: |
Z. Miskolczy, L. Biczók, SupraBank 2026, Photochromism of a Merocyanine Dye Bound to Sulfonatocalixarenes: Effect of pH and the Size of Macrocycle on the Kinetics (dataset). https://doi.org/10.34804/supra.20210928262 |
| Link: | https://doi.org/10.34804/supra.20210928262 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
Z. Miskolczy, L. Biczók, J. Phys. Chem. B 2013, 117, 648–653. |
| Link: | https://doi.org/10.1021/jp310167j |
| Export: | BibTex | RIS | EndNote | |
Binding Isotherm Simulations
The plot depicts the binding isotherm simulation of a 1:1 interaction of trans-Merocyanine (0.005714285714285714 M) and sCx4 (0 — 0.011428571428571429 M).
Please sign in: customize the simulation by signing in to the SupraBank.