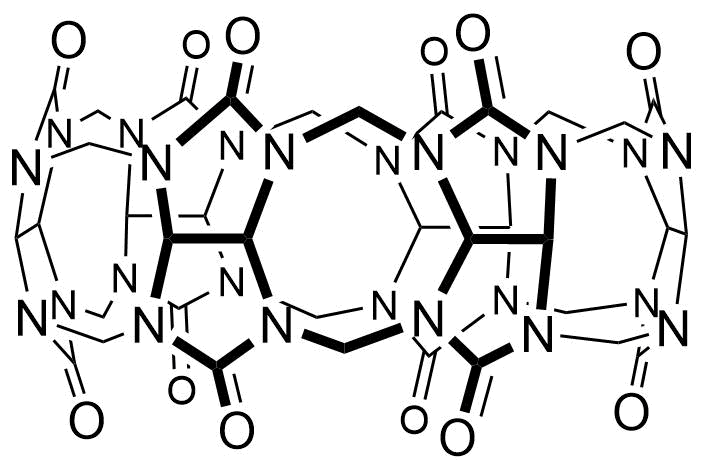
Binding Properties
| 𝜈 | Molecule 1 : 2 Host | ||
| Ka = | 5.13⋅105 | ± 2.36⋅104 | M-2 |
| Kd = | |||
| logKa = | |||
| T | 30.0 °C | ||
| Energy | kJ mol-1 | kcal mol-1 | |||
|---|---|---|---|---|---|
| ΔG | = | -33.14 | ± 0.12 | -7.92 | ± 0.03 |
| ΔH | = | -63.64 | ± 0.04 | -15.21 | ± 0.01 |
| -TΔS | = | 30.0 | ± 0.1 | 7.17 | ± 0.02 |
| J mol-1 K-1 | cal mol-1 K-1 | ||||
| ΔS | = | -99.0 | ± 0.3 | -23.7 | ± 0.1 |
These are the specifications of the determination of the experimental results.
| Detection Method: | Direct | ||
| Assay Type: | Direct Binding Assay | ||
| Technique: | Isothermal Titration Calorimetry | ||
| Instrument: | VP-ITC,MicroCal | ||
| VCell | = | 1400.0 𝜇L | |
| cmolecule | = | 500.0 𝜇M syringe | |
| cpartner | = | 130.0 𝜇M cell | |
Detailed information about the solvation.
| Solvent System | Buffer System | 10 mM ammonium chloride pH-4.0 |
| Solvents | water | |
| Additives | AMMONIUM CHLORIDE | 10.0 mM |
| Source of Concentration | estimated | |
| Total concentration | 10.0 mM | |
| pH | 4.0 |
Please find here information about the dataset this interaction is part of.
| Citation: |
E. Keinan, O. Reany, M. K. Sinha, M. Yefet, M. Botoshansky, SupraBank 2025, Bistable Cucurbituril Rotaxanes Without Stoppers (dataset). https://doi.org/10.34804/supra.20240314528 |
| Link: | https://doi.org/10.34804/supra.20240314528 |
| Export: | BibTex | RIS | EndNote |
Please find here information about the scholarly article describing the results derived from that data.
| Citation: |
M. K. Sinha, O. Reany, M. Yefet, M. Botoshansky, E. Keinan, Chemistry A European J 2012, 18, 5589–5605. |
| Link: | https://doi.org/10.1002/chem.201103434 |
| Export: | BibTex | RIS | EndNote |
Binding Isotherm Simulations
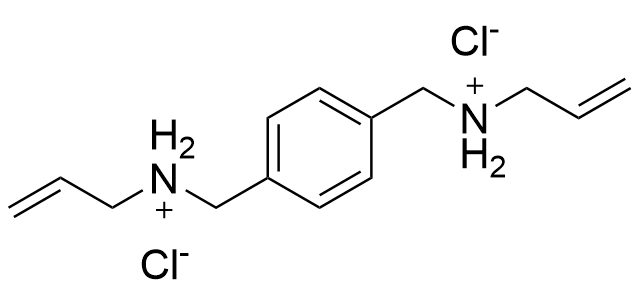
The plot depicts the binding isotherm simulation of a 1:1 interaction of N,N'-(1,4-phenylenebis(methylene))bis(prop-2-en-1-aminium) chloride (3.899395593682979e-05 M) and CB6 (0 — 7.798791187365959e-05 M).
Please sign in: customize the simulation by signing in to the SupraBank.